While studying political science at Stanford, I enrolled in a physiology course taught by the father of palmar cooling, Professor Craig Heller. In the final lecture, Professor Heller presented his research he conducted to first demonstrate how proper thermoregulation — achieved by precisely cooling the palms of the hands — could improve strength, endurance, and muscle recovery, and concluded by showcasing the
CoolMitt, an electrical palmar cooling system he helped design and recently commercialize. Wanting to enjoy these benefits, I asked Professor Heller if I could buy one; however, he informed me that CoolMitt cost thousands and was backordered to a waitlist. Having neither the money nor the patience needed for a CoolMitt, I designed an affordable palmar cooling system for myself and my friends — The Thermoroid.
Necessity is the mother of invention.
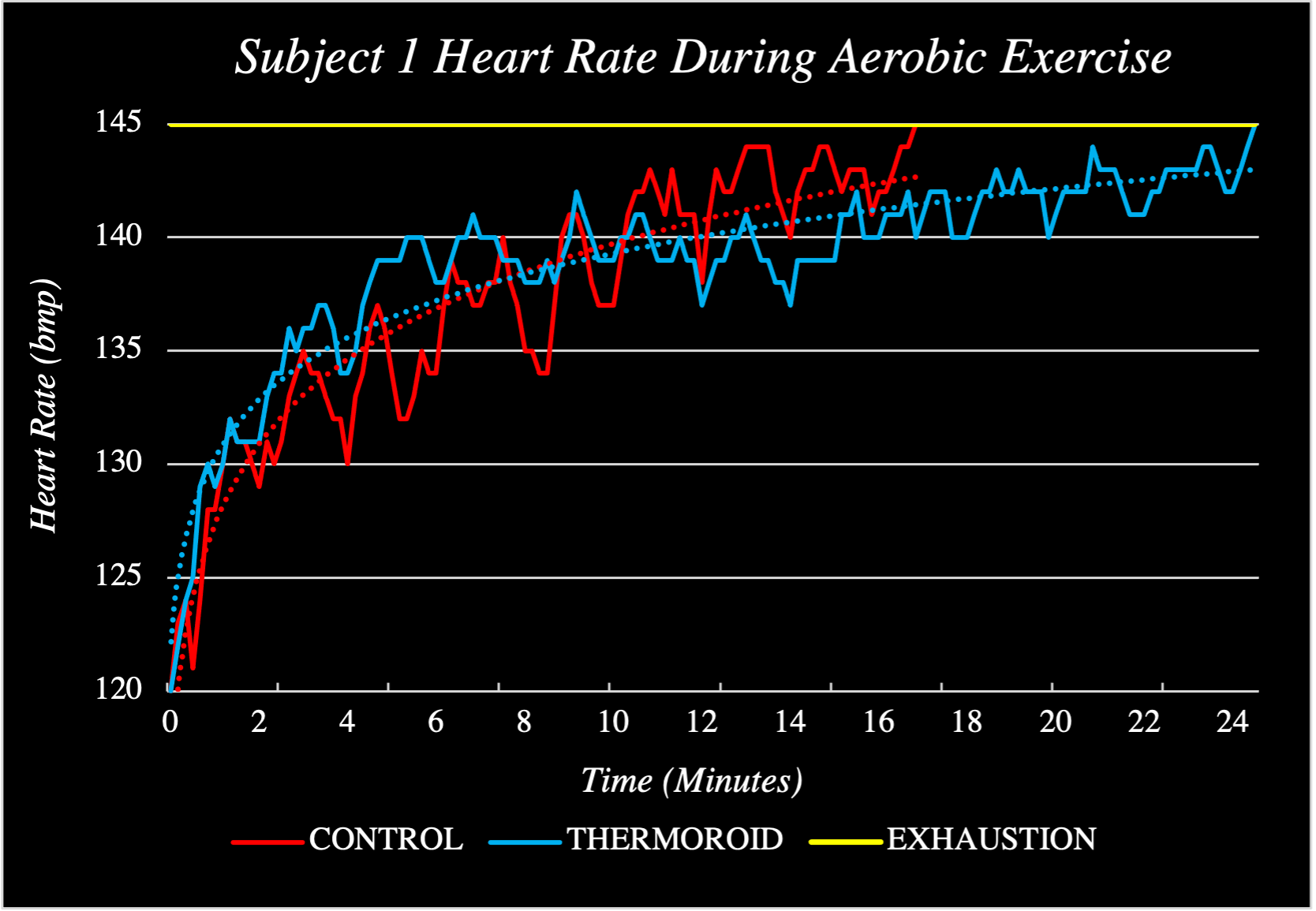
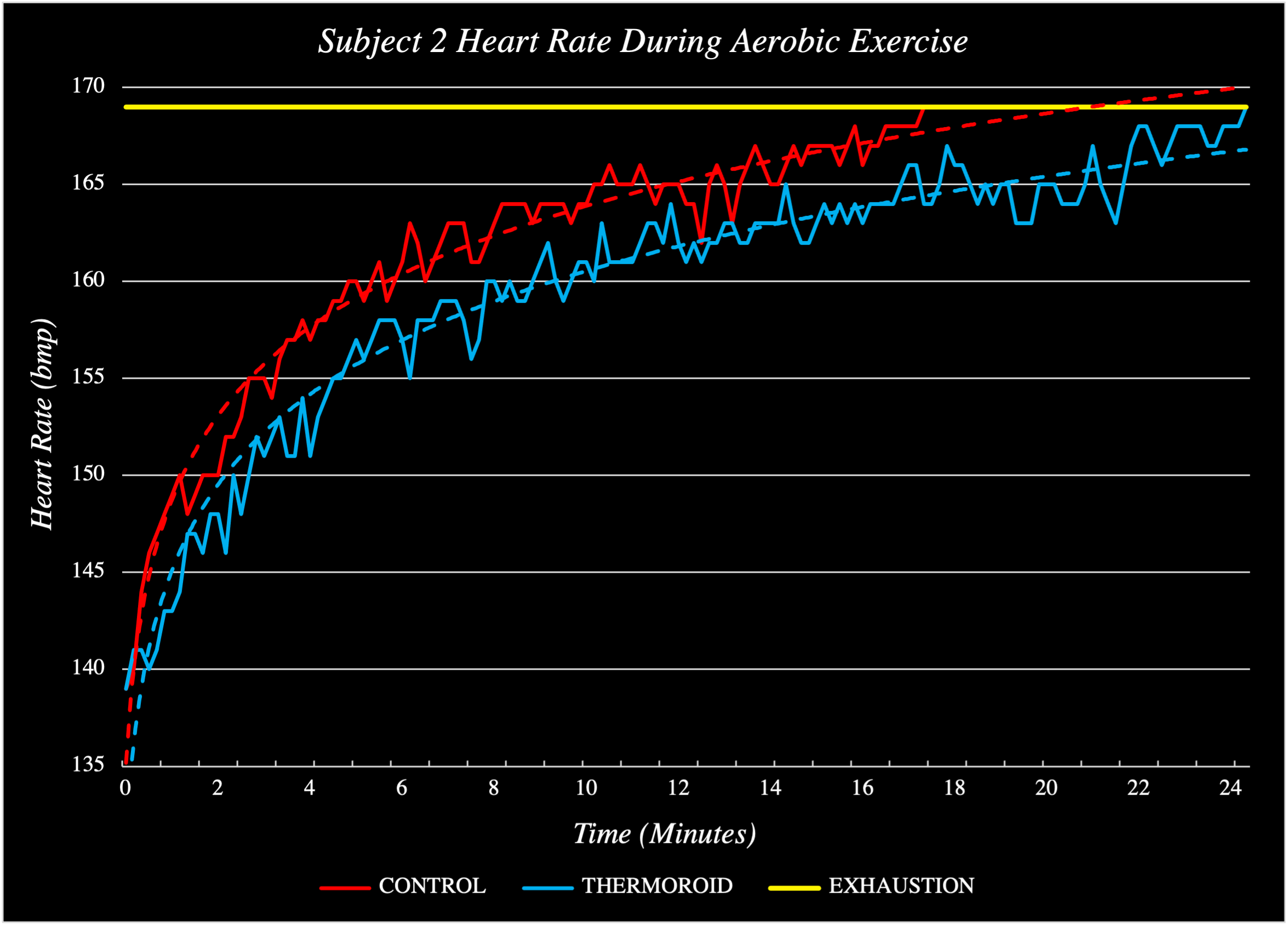
To economize palmar cooling technology, I removed the need for electrical cooling techniques and opted for a chemical solution instead. After a few weeks of researching and experimenting with various heatsinks in my dorm room, I developed a material that could absorb significant heat while remaining within the range of temperatures ideal for palmar cooling as well as change color when to indicate its temperature. I packaged this material into a handheld device, analyzed its performance, iterated its design (current prototype count: 237) and collected pilot human trials data (see: below). In Summer 2023, I filed a provisional patent on the technology and a non-provisional in October 2023. This intellectual property was acquired by biotech start-up, Arteria Technology, where I now serve as Chief Innovation Officer, working to bring the Thermoroid to market.